Does Magnesium Chloride Rust Cars?
Many people wonder if magnesium chloride causes cars to rust. This is a common question, especially for those living in areas where it’s used to de-ice roads. It can be a bit confusing because salt is known to cause rust.
But don’t worry! We’ll break down exactly what you need to know about Does Magnesium Chloride Rust Cars? in a super simple way. Stick around, and we’ll show you exactly how it works and what you can do.
Magnesium Chloride And Car Rust Facts
This section will explore the direct relationship between magnesium chloride and the common problem of car rust. We will look at why this substance is used, how it interacts with metal, and what makes it different from other de-icing agents. Understanding these points is key to knowing whether your car is truly at risk.
We aim to clear up any confusion and provide straightforward answers.
What Is Magnesium Chloride?
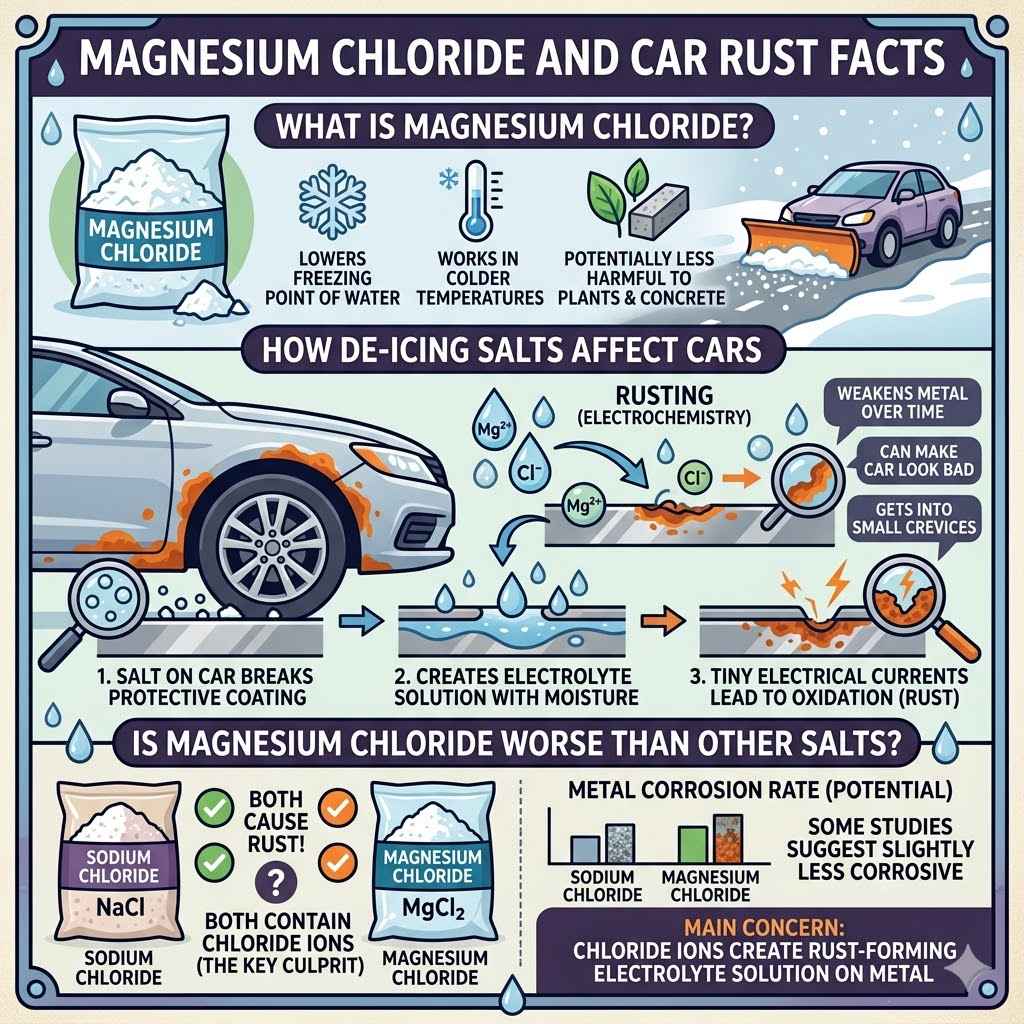
Magnesium chloride is a chemical compound. It is a type of salt that comes from the element magnesium. It looks like white crystals or flakes.
It is often used in winter. People spread it on roads to melt ice and snow. This is because it can lower the freezing point of water.
This helps keep roads safe for driving.
Magnesium chloride is popular for a few reasons. It works at colder temperatures than some other salts. It is also seen as less harmful to plants and concrete than rock salt.
This makes it a more eco-friendly choice for some.
How De-Icing Salts Affect Cars
De-icing salts, including magnesium chloride, can indeed cause rust on cars. This happens through a process called electrochemistry. Metal, especially iron in your car’s steel, can corrode when exposed to moisture and electrolytes.
Electrolytes are substances that can conduct electricity when dissolved in water.
When salt dissolves in water on your car, it creates an electrolyte solution. This solution speeds up the rust-forming process. The salt breaks down the protective coatings on your car.
It also creates tiny electrical currents between different parts of the metal. These currents cause the iron to oxidize, which is what rust is.
The longer salt sits on your car, the more damage it can do. It can get into small cracks and crevices. This makes it hard to remove completely.
Rust can weaken the metal over time. It can also make your car look bad.
Is Magnesium Chloride Worse Than Other Salts?
This is a crucial question for car owners. Many people assume all salts are equally bad. However, different salts have different chemical properties.
These properties affect how they interact with metals and the environment.
Magnesium chloride is known to be a bit different from common rock salt, which is sodium chloride. While both are electrolytes and can cause rust, the rate and severity can vary. Some studies suggest that magnesium chloride might be slightly less corrosive to metals than sodium chloride.
However, it is still a salt.
The main concern is the presence of chloride ions. These ions are the key players in the electrochemical process that leads to rust. Magnesium chloride contains a high concentration of chloride ions, just like sodium chloride.
So, while the magnesium itself might have some minor effects, the chloride is the primary culprit.
The Science Behind Rust And Magnesium Chloride
To truly understand Does Magnesium Chloride Rust Cars?, we need to look at the science. This section will break down the chemical reactions involved. We will explain how salts act as catalysts for corrosion and what role magnesium chloride plays in this process.
You’ll learn about electrochemistry in simple terms.
Electrochemistry Of Rusting
Rusting is an electrochemical process. It happens when iron reacts with oxygen and water. Think of it like a tiny battery forming on the surface of your car’s metal.
This “battery” needs a few things to work.
First, you need iron. Your car is made of steel, which is mostly iron. Second, you need oxygen from the air.
Third, you need an electrolyte. This is where salt comes in. Water alone can cause rust, but it’s a very slow process.
Electrolytes make it happen much faster.
An electrolyte is a substance that dissolves in water and creates ions. Ions are electrically charged particles. These ions allow the water to conduct electricity.
This electrical flow is what drives the chemical reaction that turns iron into rust.
How Chloride Ions Cause Corrosion
Chloride ions (Cl-) are the main reason why salts cause rust. When magnesium chloride dissolves in water, it breaks apart into magnesium ions (Mg2+) and chloride ions (Cl-). It’s these chloride ions that are very aggressive towards metal.
Chloride ions disrupt the protective oxide layer that might form on steel. This layer normally acts as a shield against further corrosion. When the chloride ions break down this shield, they expose the bare metal underneath.
This allows the rust-forming process to begin and continue.
The more chloride ions present, the faster the corrosion can occur. This is why even small amounts of salt can significantly accelerate rusting. They make the metal more susceptible to oxidation.
Magnesium Chloride Versus Other De-Icers
Let’s compare magnesium chloride to other common de-icing agents to see how it stacks up. While all salts can contribute to rust, the exact impact can differ.
Sodium chloride (rock salt) is the most common and cheapest de-icer. It’s effective at melting ice but is also known to be quite corrosive. Calcium chloride is another option.
It works at even colder temperatures than magnesium chloride. It can also be quite corrosive, and it’s known for melting ice very quickly.
Magnesium chloride is often seen as a middle ground. It works at low temperatures and is considered somewhat less corrosive than sodium chloride in some tests. However, it still contains those damaging chloride ions.
So, the difference in corrosiveness might not be enough to make it completely safe for your car.
Here’s a simple way to think about it:
- Sodium Chloride (Rock Salt): Very common, effective, quite corrosive.
- Calcium Chloride: Works at very low temperatures, melts ice fast, also corrosive.
- Magnesium Chloride: Works at low temperatures, often considered less corrosive than rock salt, but still contains chloride ions and can cause rust.
Practical Steps To Prevent Car Rust
Now that we know the science, let’s focus on what you can do. This section provides actionable advice. We will cover washing your car regularly, using protective coatings, and inspecting for damage.
These steps are essential for anyone concerned about Does Magnesium Chloride Rust Cars? and its impact.
The Importance Of Regular Car Washing
Washing your car frequently is one of the most effective ways to combat rust. When de-icing salts are present, they stick to the underside and body of your vehicle. These salts, including magnesium chloride, create a corrosive environment.
A good car wash removes these salt residues. It’s especially important to wash the undercarriage. This is where most of the salt spray collects from the road.
Washing should be done after driving on treated roads, particularly during winter months.
When you wash, use a strong stream of water. Consider using an undercarriage spray attachment if you have one. This helps to dislodge and wash away the salt and grime effectively.
Even a simple rinse can make a big difference in preventing long-term damage.
Protective Coatings And Treatments
Beyond regular washing, you can add extra layers of protection. These coatings create a barrier between the salt and your car’s metal. This barrier helps to prevent the corrosive elements from reaching the steel.
Undercoating is a common option. This is a protective layer applied to the underside of your car. It’s made of various materials, like rubberized asphalt or wax-based compounds.
This layer shields the metal from salt, moisture, and road debris.
Rustproofing is another service. This often involves spraying a protective substance into the cavities of your car, like door panels and frame rails. These areas are prone to rust from the inside out.
Even applying a good quality wax or sealant to your car’s paintwork can help. While it’s primarily for paint protection, it can offer some minor resistance against salt corrosion.
Inspecting And Repairing Damage Early
Rust doesn’t appear overnight. It starts small and grows over time. Catching rust spots early is key to stopping them from spreading.
Regularly inspect your car for any signs of rust.
Pay close attention to areas prone to rust. These include wheel wells, rocker panels (along the bottom of the doors), door edges, and the frame. Look for any bubbling paint or small rust spots.
If you find a small rust spot, get it repaired as soon as possible. A small spot can be sanded down, treated with a rust inhibitor, and repainted. If you let it grow, it can become a much larger and more expensive repair.
It can even compromise the structural integrity of your car.
Here are some key areas to check:
- Wheel Wells: These areas get a lot of road spray and debris.
- Rocker Panels: The metal strips below the doors are magnets for salt and moisture.
- Door Edges and Bottoms: Water and salt can collect and sit here, causing rust.
- Bumpers and Lower Body Panels: Especially if they are made of metal, these areas are exposed.
- Frame and Undercarriage: Essential structural parts that need regular inspection.
Real-Life Examples And Scenarios
To make the information more concrete, let’s look at some real-world examples. These stories show how magnesium chloride affects cars and what people have done. They help answer Does Magnesium Chloride Rust Cars? with practical evidence.
Case Study A A Commuter In A Snowy Climate
Sarah lives in a city that frequently uses magnesium chloride for de-icing. She drives her car to work every day during the winter. She used to only wash her car a couple of times a year.
After a few winters, she noticed rust spots appearing on her car’s fenders and along the bottom of the doors. Her mechanic told her that the salt spray from the roads was the main cause. He explained how the magnesium chloride was attacking the metal.
Sarah decided to change her routine. She started washing her car every two weeks during the winter. She also got an undercoating treatment done at the beginning of the season.
Over the next few years, she saw a significant reduction in new rust spots. Her car looked much better, and the repairs were less extensive.
Case Study B A Car Owner Who Didn’t Wash
John lives in a rural area where magnesium chloride is used liberally on roads. He doesn’t have a regular car wash nearby and rarely washes his car, especially in winter. His car is older, and he figures some rust is inevitable.
After about five years of living in this climate, John’s car started to show serious rust. The frame had significant corrosion, and there were holes forming in the rocker panels. He also noticed rust coming through on the trunk lid.
When he took it to a body shop, they told him the rust was so bad that it would cost more to repair than the car was worth. The magnesium chloride had attacked the metal relentlessly because it was never washed away. This is a prime example of what happens when you ignore the salt.
Scenario 1 Driving In A Salted Area
Imagine you are driving on a highway right after a snowstorm. The road crews have applied magnesium chloride to melt the ice. As you drive, tiny particles of the de-icer spray up from the road.
These particles hit the underside of your car, your wheel wells, and the lower parts of your doors and fenders. They mix with any moisture already present on your car. This creates a corrosive solution that begins to eat away at the metal’s protective coatings.
If you don’t wash your car soon, these salt residues will sit there for days or weeks. They will continue to cause damage. The longer they stay, the deeper the rust can penetrate.
Scenario 2 Storing A Car In Winter
Let’s say you have a classic car or a vehicle you don’t use in the winter. You might store it in an unheated garage or outdoors. Before storing it, you drive it on roads that have been treated with magnesium chloride.
Even though the car won’t be driven, the salt that is already on it will remain. If the car is not thoroughly washed and dried before storage, the salt will continue to react with the metal. Moisture trapped on the surface can exacerbate the problem.
This is why it’s vital to wash your car thoroughly, especially the undercarriage, before putting it away for an extended period, even if it’s not being driven. Proper cleaning before storage prevents the salt from causing unseen damage over time.
Statistics And Expert Opinions
To further support our understanding of Does Magnesium Chloride Rust Cars?, let’s look at some data. Statistics and expert views help paint a clearer picture of the issue.
Corrosion Rates
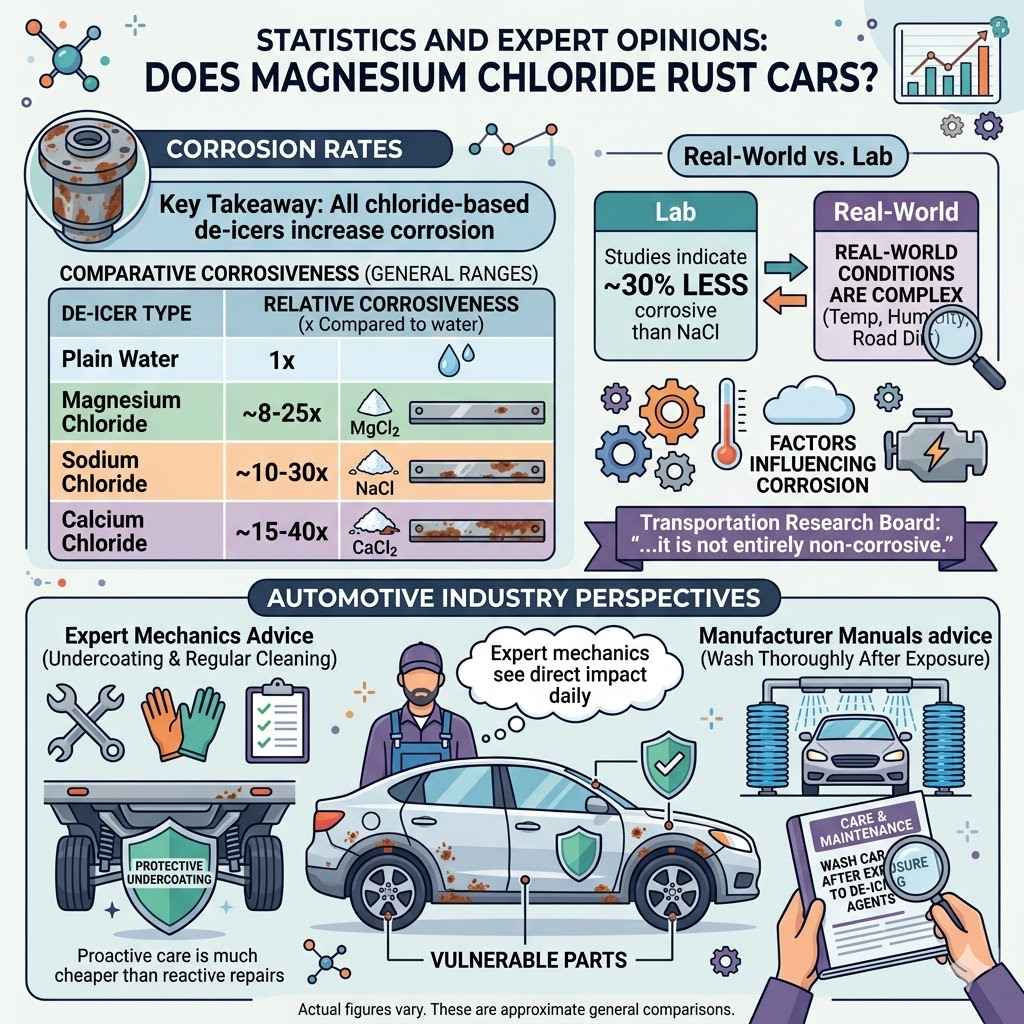
Studies on de-icing salts and vehicle corrosion show varied results. However, a general consensus exists that all chloride-based de-icers increase the rate of corrosion on vehicles.
Some research has indicated that magnesium chloride might be up to 30% less corrosive than sodium chloride under specific laboratory conditions. However, real-world conditions are much more complex. Factors like temperature, humidity, and road dirt can influence corrosion rates significantly.
A study by the Transportation Research Board noted that while magnesium chloride might offer some benefits, “it is not entirely non-corrosive.” This means that while it might be a slightly better option, it’s still a salt and poses a rust risk.
Here’s a general comparison often seen in studies, though actual figures can vary widely based on testing methods:
| De-Icer Type | Relative Corrosiveness (Compared to plain water) |
|---|---|
| Plain Water | 1x |
| Sodium Chloride | ~10-30x |
| Calcium Chloride | ~15-40x |
| Magnesium Chloride | ~8-25x |
These numbers are approximate and can change. The key takeaway is that all these salts significantly increase corrosion.
Automotive Industry Perspectives
Automotive manufacturers and repair shops are well aware of the damage caused by road salts. They often advise car owners to take preventative measures, especially in regions with harsh winters.
Many car manuals will include sections on care and maintenance that specifically mention the need to wash the vehicle thoroughly after exposure to de-icing agents. They understand the chemical properties of these salts and their potential to cause harm.
Expert mechanics frequently recommend undercoating and regular cleaning. They see the direct impact of salt corrosion daily. Their advice focuses on practical steps owners can take to prolong the life of their vehicles.
They stress that proactive care is much cheaper than reactive repairs.
Frequently Asked Questions
Question: Does magnesium chloride damage car paint?
Answer: Yes, like other de-icing salts, magnesium chloride can damage car paint if left on for too long. The salt and moisture can break down the paint’s protective layers, leading to dullness, fading, or even peeling in severe cases. Regular washing is key to preventing this.
Question: Can I use a pressure washer to clean salt off my car?
Answer: Yes, a pressure washer can be very effective for removing salt. Just be careful not to use too high a pressure or spray too close to sensitive areas like seals or plastic trim, as this could cause damage. Focus on the undercarriage and wheel wells.
Question: How often should I wash my car in winter if magnesium chloride is used?
Answer: If you drive on roads treated with magnesium chloride, it’s recommended to wash your car at least every two weeks during the winter. If you drive frequently on treated roads or during heavy salt application, consider washing it weekly.
Question: Is magnesium chloride bad for electric cars?
Answer: While electric cars have different components than gasoline cars, the metal body and frame are still susceptible to rust. So, magnesium chloride can still cause corrosion on electric vehicles just as it can on traditional cars. Regular washing is still important.
Question: What is the best way to remove magnesium chloride residue?
Answer: The best way to remove magnesium chloride residue is through thorough washing with water and a car soap designed to lift road grime. Ensure you clean the undercarriage, wheel wells, and any other areas where salt spray might accumulate. A good rinse and drying can also help.
Summary
Yes, magnesium chloride can cause cars to rust. While it might be slightly less corrosive than rock salt in some tests, it still contains damaging chloride ions. These ions accelerate the rusting process on your car’s metal.
Protecting your vehicle involves regular washing, applying protective coatings, and promptly repairing any signs of rust.







