What Causes Car Battery Terminals to Corrode Fast
Many car owners find themselves wondering, “What Causes Car Battery Terminals to Corrode Fast?” It’s a common puzzle, especially if you’re new to car care. You see that white or bluish gunk building up, and it makes you worry about your car’s battery. Don’t fret!
This problem is easily understood and solved. We’ll walk through it simply, step by step, so you can fix it and keep your car running smoothly. Let’s find out what’s causing that corrosion.
Understanding Battery Terminal Corrosion
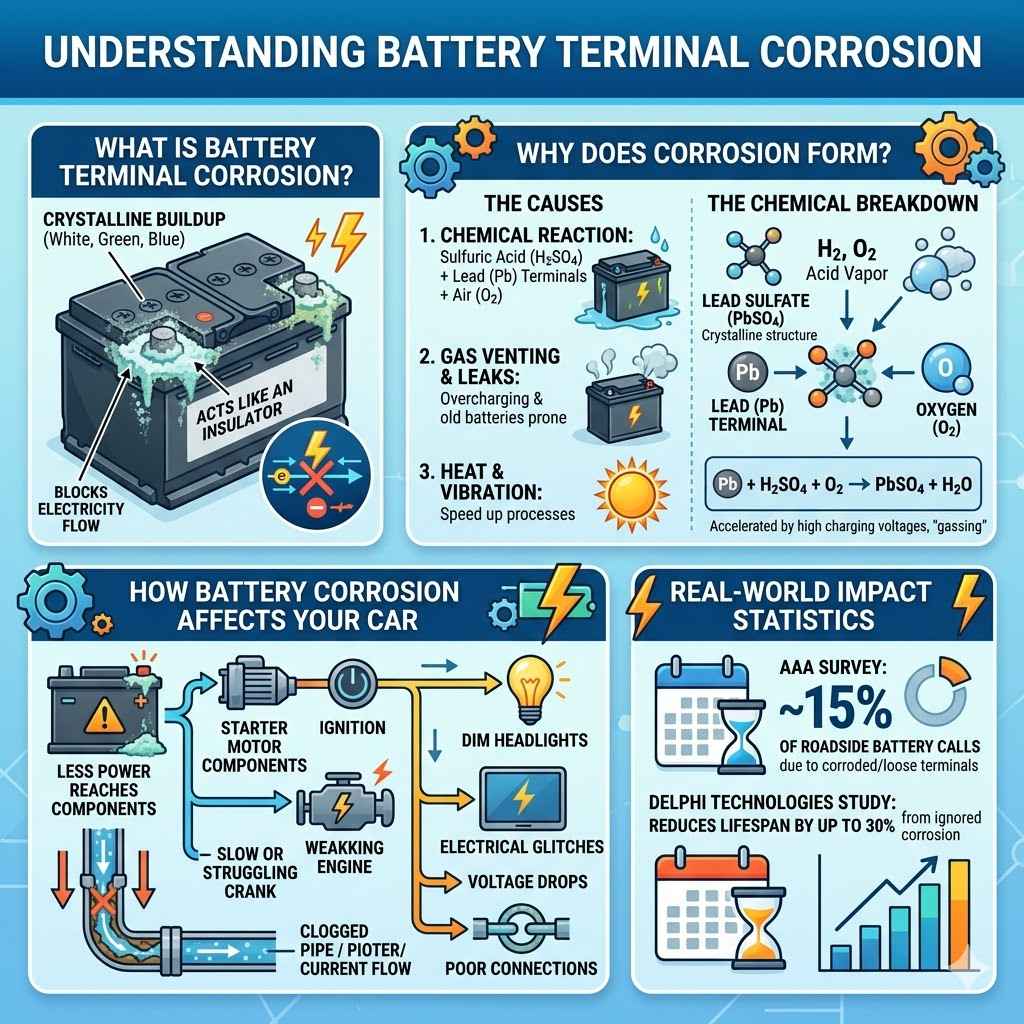
What is Battery Terminal Corrosion?
Battery terminal corrosion is that fuzzy, powdery stuff that appears on your car battery’s posts and terminals. It’s usually white, greenish, or bluish. This buildup isn’t just ugly; it’s a real problem for your car’s electrical system.
It acts like an insulator, blocking the flow of electricity between the battery and your car. This can lead to starting issues, dim lights, or other electrical glitches. It happens when the battery acid leaks out a little and reacts with the metal of the terminals.
Why Does Corrosion Form?
The main reason corrosion forms is a chemical reaction. When the sulfuric acid inside your battery escapes, even in tiny amounts, it can mix with the air and the lead terminals. This combination creates lead sulfate, which is what you see as corrosion.
Overcharging your battery can also make it vent more gas, increasing the chances of this reaction. Old batteries are more prone to leaking, so they often show corrosion sooner. Heat can also speed up these chemical processes, making corrosion worse in hot climates.
The Chemical Breakdown
At its heart, battery terminal corrosion is a byproduct of electrochemistry. The lead and lead dioxide within the battery react with sulfuric acid during discharge to produce electricity. However, this process isn’t perfectly sealed.
Small amounts of hydrogen gas and acid vapor can escape from the battery, especially when it’s under stress or aging. When these vapors meet the atmospheric oxygen and the lead terminals, a slow oxidation reaction occurs. This forms a crystalline structure, primarily lead sulfate, which is the visible corrosion.
The chemical formula involved is complex, but a simplified view shows lead (Pb) reacting with sulfuric acid (H2SO4) and oxygen (O2) to form lead sulfate (PbSO4) and water (H2O). This process is accelerated by heat, vibration, and poor ventilation. The resulting lead sulfate is an electrical insulator, hence the problems it causes.
High charging voltages can also lead to “gassing,” where the battery actively releases hydrogen and oxygen, carrying acid mist with it, which further contributes to terminal corrosion.
How Battery Corrosion Affects Your Car
Corrosion creates a barrier that hinders electrical conductivity. This means less power can reach your car’s starter motor, ignition system, or other electronics. You might notice your engine cranks slowly or struggles to start.
Your headlights might appear dim, or other electrical components might not work correctly. In severe cases, the corrosion can eat away at the terminals themselves, leading to a poor connection that can cause intermittent electrical failures. It’s like trying to push water through a clogged pipe; the flow is restricted.
A corroded terminal can create resistance in the electrical circuit. This resistance causes voltage drops, meaning the components that need a certain voltage to operate won’t receive it. For instance, the starter motor needs a significant amount of current to turn the engine over.
If the terminals are corroded, this current flow is impeded, leading to a weak or non-existent crank. Similarly, sensors and computers in your car require stable voltage to function properly. Corrosion can disrupt this stability, leading to error codes or erratic behavior.
Real-World Impact Statistics
According to a survey by AAA, approximately 15% of roadside assistance calls for battery-related issues are due to corroded terminals or loose connections. This translates to millions of drivers experiencing inconvenience and potential repair costs each year. Furthermore, a study by Delphi Technologies found that ignoring minor corrosion can lead to premature battery failure, reducing a battery’s lifespan by up to 30%.
Common Causes of Fast Battery Terminal Corrosion
Battery Age and Condition
Older batteries are naturally more likely to corrode. As a battery ages, its internal components degrade, and it becomes more prone to leaking small amounts of acid or gas. This leakage is a primary driver of corrosion.
A battery that is nearing the end of its service life might not hold a charge as well and could be subjected to more frequent overcharging cycles, both of which contribute to corrosion. Even a battery that looks fine might have internal issues causing it to vent gases.
The materials used in battery construction also wear down over time. The lead plates can become brittle, and the seals that contain the electrolyte might weaken. This allows the acidic solution to escape more easily.
A battery that has been deeply discharged multiple times or has been exposed to extreme temperatures also suffers from accelerated aging. These factors combine to create a perfect storm for corrosion to develop rapidly on the terminals.
Battery Overcharging
When your car’s charging system, controlled by the alternator and voltage regulator, sends too much voltage to the battery, it causes overcharging. This excess voltage forces the battery to work too hard, generating excessive heat and releasing hydrogen gas. This gas, along with any escaping acid mist, is a major culprit in accelerating terminal corrosion.
The battery essentially boils internally, venting harmful fumes.
A faulty voltage regulator is a common cause of overcharging. It’s supposed to stop sending power to the battery once it’s full. If it malfunctions, it can keep pushing current into a full battery.
This can damage the battery plates and lead to the venting of gases. You might notice a sulfuric smell coming from your engine bay if your battery is being overcharged. This strong smell is a clear indicator that something is wrong with the charging system.
Battery Undercharging
Conversely, undercharging can also contribute to corrosion, though in a slightly different way. When a battery is consistently undercharged, it doesn’t reach its full capacity, and some of the lead sulfate crystals formed during discharge may not be converted back to lead and lead dioxide during charging. These “hardened” lead sulfate crystals can impede the flow of electricity.
More importantly, incomplete charging can lead to uneven chemical reactions within the battery, potentially causing localized acid buildup and venting.
An undercharged battery is also more susceptible to sulfation, a process where lead sulfate hardens and coats the battery plates. This reduces the battery’s ability to accept a charge and deliver power. While sulfation is different from terminal corrosion, the conditions that cause undercharging (like a weak alternator or frequent short trips where the battery doesn’t get enough time to recharge) can also lead to other battery issues that indirectly promote corrosion.
The battery is under stress, increasing the likelihood of venting.
Extreme Temperatures
Both very hot and very cold temperatures can negatively impact your car battery and lead to faster corrosion. In hot weather, batteries work harder, and chemical reactions speed up. This increased activity can lead to more gassing and acid evaporation.
Heat also degrades the internal components of the battery more quickly, making it more prone to leakage. A hot battery is essentially an overworked battery.
In cold weather, the chemical reactions inside the battery slow down. This means the battery has to work harder to produce the same amount of power. If the battery is already weak, extreme cold can cause it to freeze if it’s not sufficiently charged.
Cold also thickens the oil in your engine, making it harder to crank, putting more strain on the battery. This added strain, coupled with the battery’s reduced efficiency in the cold, can lead to more frequent deep discharges, which in turn can accelerate corrosion when warmer weather returns.
A battery operating in an engine bay that frequently experiences temperatures above 100°F (38°C) can have its lifespan reduced by half. The heat accelerates the natural degradation of the battery’s internal materials. Conversely, while cold slows reactions, a battery at 0°F (-18°C) can only deliver about 50% of its rated cranking power compared to a battery at 70°F (21°C).
This puts immense stress on the battery, especially during startup, potentially leading to venting or strain that indirectly promotes corrosion.
Damaged Battery Case or Seals
If the plastic casing of your battery is cracked or damaged, or if the seals around the terminals are compromised, it can allow battery acid and gases to escape freely. This direct leakage is a very common and significant cause of rapid corrosion buildup. Even a small crack, perhaps caused by a loose component hitting the battery or improper installation, can be enough to let corrosive materials out.
The seals are designed to keep the electrolyte contained within the battery. If these seals fail, either due to age, extreme temperature fluctuations, or physical damage, acid can seep out. This acid then reacts with the air and the terminal posts, forming corrosion.
It’s crucial to inspect the battery case for any signs of damage or leakage. Sometimes, a leaking battery needs to be replaced entirely, as the damage might be beyond simple repair.
Vibrations
Excessive vibration can jostle the battery and its connections. This constant shaking can loosen terminal clamps, allowing for air to enter and react with the metal. More importantly, significant vibrations can cause internal damage to the battery itself over time.
This can lead to micro-cracks in the casing or seals, or even damage the internal plates, increasing the likelihood of acid leakage and subsequent corrosion. A securely mounted battery is essential.
If your battery isn’t properly secured in its tray, or if your vehicle’s suspension is worn, leading to a rougher ride, the battery can be subjected to constant jolting. This can accelerate wear and tear on the battery’s internal structure. Think of it like shaking a carton of milk vigorously; eventually, it might start to leak.
The same principle applies to a car battery under constant vibration. This constant movement can also cause the metal terminals to shift slightly, creating tiny gaps for air to ingress.
Charging System Issues (Alternator/Regulator)
As mentioned earlier, problems with the alternator or voltage regulator are significant contributors to corrosion. If the alternator is faulty and not producing the correct voltage, it can lead to either undercharging or overcharging the battery. An overcharging system is particularly bad, as it forces the battery to vent harmful gases and heat up excessively, directly promoting corrosion.
The voltage regulator’s job is to maintain a steady, appropriate voltage, usually around 13.5 to 14.5 volts.
A weak alternator might struggle to keep the battery fully charged, especially during periods of high electrical demand (like driving at night with headlights, wipers, and A/C on). This can lead to a chronically undercharged battery. A failing voltage regulator, on the other hand, might allow the alternator to over-speed and over-charge the battery.
This overcharging generates excessive heat and leads to the release of hydrogen gas, which mixes with battery acid vapor to create corrosive buildup. It’s a delicate balance that, when disrupted, quickly causes problems.
Short Trips and Infrequent Driving
If you mostly take short trips, your battery might not get enough time to fully recharge. A car’s alternator needs a certain amount of driving time at consistent speeds to replenish the power used to start the engine and run accessories. Short trips mean the alternator is constantly playing catch-up.
This leads to a chronically undercharged battery. As discussed, an undercharged battery can lead to sulfation and other issues that indirectly contribute to corrosion.
Infrequent driving presents a similar problem. When a car sits for long periods, the battery slowly discharges itself due to the car’s electronic systems (alarms, clock, computer memory). If the battery isn’t being recharged regularly, it can become deeply discharged.
This prolonged low state of charge can cause irreversible sulfation and damage, making the battery more susceptible to venting and corrosion when it’s finally put to use or charged. It’s like leaving a phone unplugged for months; its ability to hold a charge will be compromised.
How to Prevent and Clean Battery Terminal Corrosion
Regular Inspection
The best way to combat corrosion is to catch it early. Make it a habit to visually inspect your battery terminals every few months, especially during routine maintenance like oil changes. Look for any signs of white, blue, or greenish powdery buildup around the battery posts and cable clamps.
Also, check that the battery itself is securely fastened in its tray and that there are no cracks in the battery case.
A quick visual check can save you a lot of trouble down the line. If you notice even a small amount of corrosion, it’s easier to clean and prevent it from returning. Think of it like checking for a small leak in your roof; fixing it when it’s small is much cheaper and easier than waiting until the whole ceiling is ruined.
This proactive approach keeps your electrical system healthy.
Cleaning Corroded Terminals
To clean corroded terminals, you’ll need a few basic items: safety glasses, gloves, a wire brush (or a specific battery terminal cleaning tool), baking soda, water, and some petroleum jelly or dielectric grease. First, disconnect the battery cables, starting with the negative (black) terminal, then the positive (red). This prevents accidental shorts.
Mix a solution of baking soda and water (about a tablespoon of baking soda per cup of water). This alkaline solution will neutralize the acid. Apply this mixture to the corroded terminals and posts using an old toothbrush or a rag.
You’ll see it fizz as it neutralizes the acid. Let it sit for a few minutes. Then, use a wire brush or terminal cleaning tool to scrub away the loosened corrosion.
Rinse with clean water and dry thoroughly with a clean cloth. Ensure no baking soda residue is left behind.
Applying Anti-Corrosion Protection
After cleaning and drying the terminals and posts, it’s crucial to apply a protective layer to prevent future corrosion. You can use a thin coating of petroleum jelly (Vaseline) or a specialized dielectric grease. This creates a barrier that prevents moisture and air from reaching the metal, significantly slowing down the corrosion process.
Ensure the cable clamps are tightened securely onto the posts after applying the grease.
These protective coatings are inexpensive and readily available at auto parts stores. They are designed to withstand the high temperatures under the hood and the harsh environment around the battery. It’s not about sealing the electrical connection tightly, but rather about creating a protective barrier on the outside of the connection point.
This simple step can make a big difference in the longevity of your battery and connections.
Securing the Battery Properly
A battery that is loose in its tray is a ticking time bomb for corrosion and other issues. Ensure the battery hold-down clamp is tight and secure. The battery should not be able to move or shift in any direction.
This prevents excessive vibration from damaging the battery and its connections, which can lead to leaks and corrosion. A firmly seated battery is essential for its longevity and the reliability of your car’s electrical system.
Check your vehicle’s manual for the correct way to secure your specific battery. Some use a clamp at the base, while others have a top strap. Over-tightening can also damage the battery case, so aim for snug and secure.
If you notice the battery tray itself is rusted or damaged, it might need to be repaired or replaced to ensure a solid mounting point. A stable battery foundation is key.
Maintaining the Charging System
Regularly check your car’s charging system to ensure it’s functioning correctly. This can be done with a voltmeter or by having a mechanic test your alternator and voltage regulator. If you suspect your battery is being overcharged or undercharged, get it diagnosed and repaired promptly.
A properly functioning charging system prevents the extreme conditions that lead to rapid corrosion.
Most auto parts stores offer free battery and charging system tests. If your car battery is consistently reading below 12.4 volts when the engine is off, or if it drops significantly below 13 volts while the engine is running, it might indicate a charging problem. Addressing these issues quickly can save your battery from premature failure and prevent corrosion issues.
Consider Battery Maintenance Products
Several aftermarket products are designed to prevent battery terminal corrosion. These include anti-corrosion sprays, terminal protectors, and terminal washers that contain anti-corrosive compounds. While a good cleaning and a layer of petroleum jelly are often sufficient, these products can offer an extra layer of protection, especially in harsh environments or for vehicles that experience a lot of vibration or temperature extremes.
These products work by creating a barrier that resists the chemical reactions that cause corrosion. Some are applied as a spray after cleaning, while others are included in felt washers that go under the terminal clamps. They are relatively inexpensive and can be a worthwhile addition to your battery maintenance routine, especially if you live in an area with high humidity or extreme temperatures.
They offer convenience and long-lasting protection.
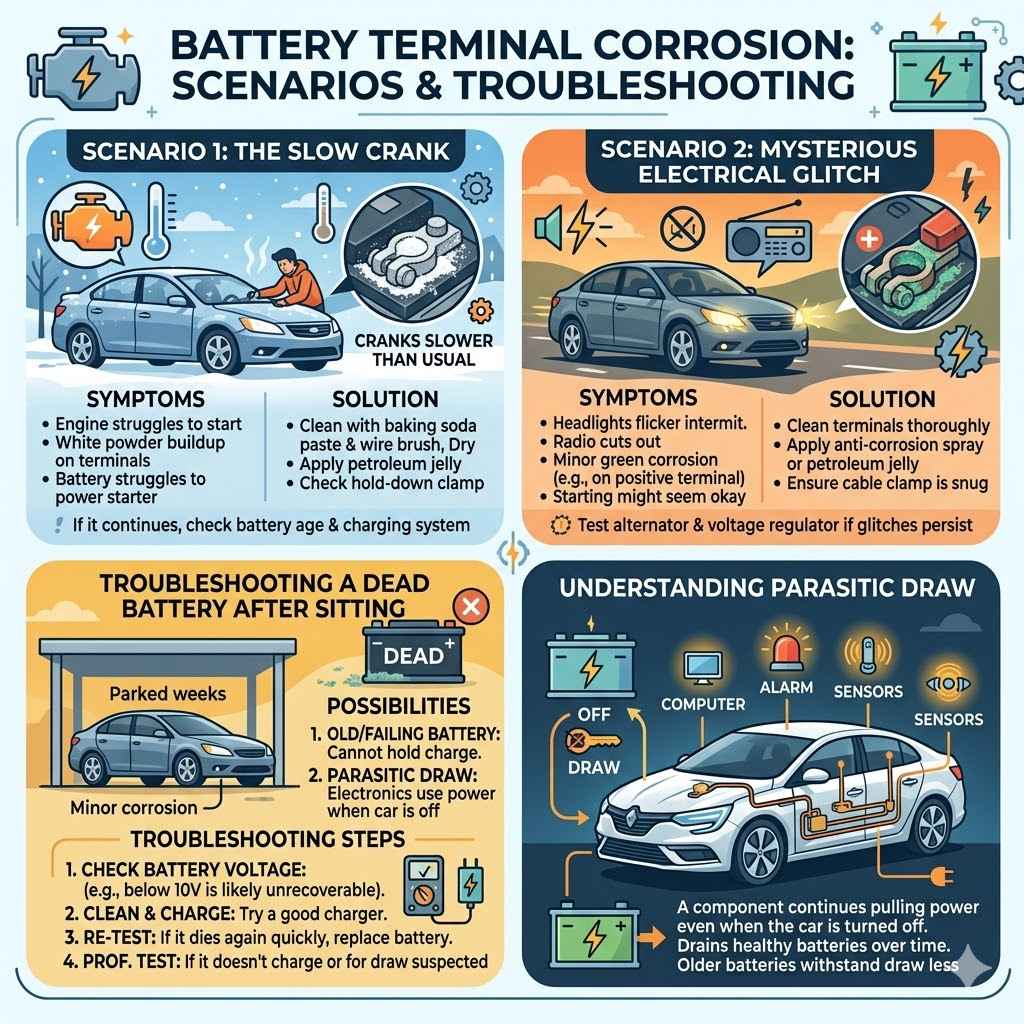
Sample Scenarios and Troubleshooting
Scenario 1 The Slow Crank
You get into your car on a cold morning, and the engine cranks much slower than usual. It eventually starts, but you’re worried. You pop the hood and see a significant amount of white, powdery buildup on your battery terminals.
This is a classic sign that corrosion is impeding the flow of electrical current to the starter motor. The slow crank is the battery struggling to send enough power through the corroded connections.
Solution: You would clean the terminals using the baking soda and water paste and a wire brush. After drying, apply petroleum jelly. Check if the hold-down clamp is tight.
If the problem persists after cleaning, the battery itself might be old or failing, or there could be an issue with the charging system.
Scenario 2 The Mysterious Electrical Glitch
Your headlights flicker intermittently, and your radio sometimes cuts out. You haven’t noticed any starting problems, so you don’t suspect the battery. However, upon closer inspection, you find a small amount of greenish corrosion on the positive battery terminal.
This corrosion, even if minor, is creating enough resistance to cause these random electrical issues. The varying electrical demands of different systems can exacerbate the problem.
Solution: Clean the terminals thoroughly. Apply anti-corrosion spray or petroleum jelly. Ensure the cable clamp is snug.
If these electrical glitches continue, it might be worth having your alternator and voltage regulator tested, as inconsistent voltage can sometimes mimic corrosion issues or be a contributing factor.
Troubleshooting a Dead Battery After Sitting
You left your car parked for a few weeks, and now it won’t start. The battery is completely dead. You might notice signs of corrosion on the terminals.
This situation points to a few possibilities: the battery could have been old and unable to hold a charge, or the car’s parasitic draw (electronics that use power when the car is off) might have drained it. The corrosion could be a symptom of the battery’s overall poor health or a cause of its inability to accept a charge.
Troubleshooting Steps:
- Check battery voltage. If it’s below 10 volts, it might be unrecoverable.
- Clean terminals and try to charge the battery with a good charger.
- If it charges but dies again quickly, the battery is likely bad.
- If it doesn’t accept a charge, or you suspect a parasitic draw, have it tested by a professional.
A parasitic draw is when a component in your car continues to pull power even when the car is off. This can drain a healthy battery over time. If the battery is old, it won’t have the reserve capacity to withstand this drain.
Both issues can lead to a dead battery and often coincide with or exacerbate corrosion problems.
Frequently Asked Questions
Question: How often should I check my car battery terminals for corrosion?
Answer: It’s a good idea to check your battery terminals at least every six months, or more frequently if you live in a hot climate or notice any unusual electrical behavior in your car.
Question: Can I use a regular metal brush to clean battery terminals?
Answer: A wire brush is suitable, but be careful not to scrub too hard, as you don’t want to damage the battery posts or terminals. A dedicated battery terminal brush is ideal.
Question: What happens if I don’t clean battery terminal corrosion?
Answer: If left untreated, corrosion builds up, restricts electrical flow, can lead to starting problems, dim lights, and eventually damage the battery and cable connections, requiring more costly repairs.
Question: Is a little bit of corrosion normal?
Answer: A very tiny, almost imperceptible amount might form over time, but any visible white, blue, or green powdery substance is a sign of a problem that should be addressed to prevent it from worsening.
Question: Can a corroded battery cause my car to not start at all?
Answer: Yes, severe corrosion can create such a strong electrical resistance that it prevents enough power from reaching the starter motor, rendering the car unable to start.
Conclusion
The primary causes of fast car battery terminal corrosion are battery age, overcharging or undercharging, extreme temperatures, and physical damage. By performing regular inspections, cleaning terminals with a baking soda solution, and applying protective grease, you can prevent this common issue. Ensuring your battery is securely mounted and your charging system functions correctly are also key steps.







